Autologous vs Non-autologous Cells Transplantation
Autologous comes from the Greek roots auto- (self) and –logos (relation). So, autologous means relative to self. The opposite term would be non-autologous. Inside non-autologous are included the terms allogeneic and xenogeneic, which are also of Greek origin. Allogeneic (allo– (other), –gen (born of, produced by), and –ic, (in the manner of)) that means from another individual of the same species, and xenogeneic (xeno-, (strange, foreign; stranger, foreigner), –gen, (born of, produced by), and –ic (in the manner of)) that means sourced from different species, including other animals, plants, or even bacteria.1
Exosomes Safety and Efficacy Profile
Autologous exosomes are associated with a favorable safety profile and a high efficacy. Due to their autologous nature, these small vesicles between 30 to 150 nm, secreted by almost all cells, do not elicit adverse effects or rejection. Furthermore, autologous exosomes do not undergo clearance by the immune system, allowing prolonged persistence at the transplantation site. On the contrary, manufactured non-autologous exosomes can present changes in their components or architectural structure that can impact their efficacy and safety.2 Furthermore, non-autologous exosomes are prone to rapid clearance, increasing with repeated administration.3
Exosomes Immune Compatibility
When autologous exosomes are transplanted, their molecular markers, such as proteins and lipids, are recognized as ‘self’ by the recipient’s immune system. This fact nullifies the risk of immune reaction (activation, inhibition, or rejection). This recognition does not happen when exosomes are sourced from different individuals of the same species—allogeneic exosomes. This is why serological, and histocompatibility tests are required for allogenic transplantations, which can only be done by authorized centers.
Exosomes sourced from other species—xenogeneic exosomes— can trigger an immune response. For this reason, their implantation is restricted to preclinical research only. The immune response includes inflammation, allergic reactions, or even severe immune-mediated damage.4
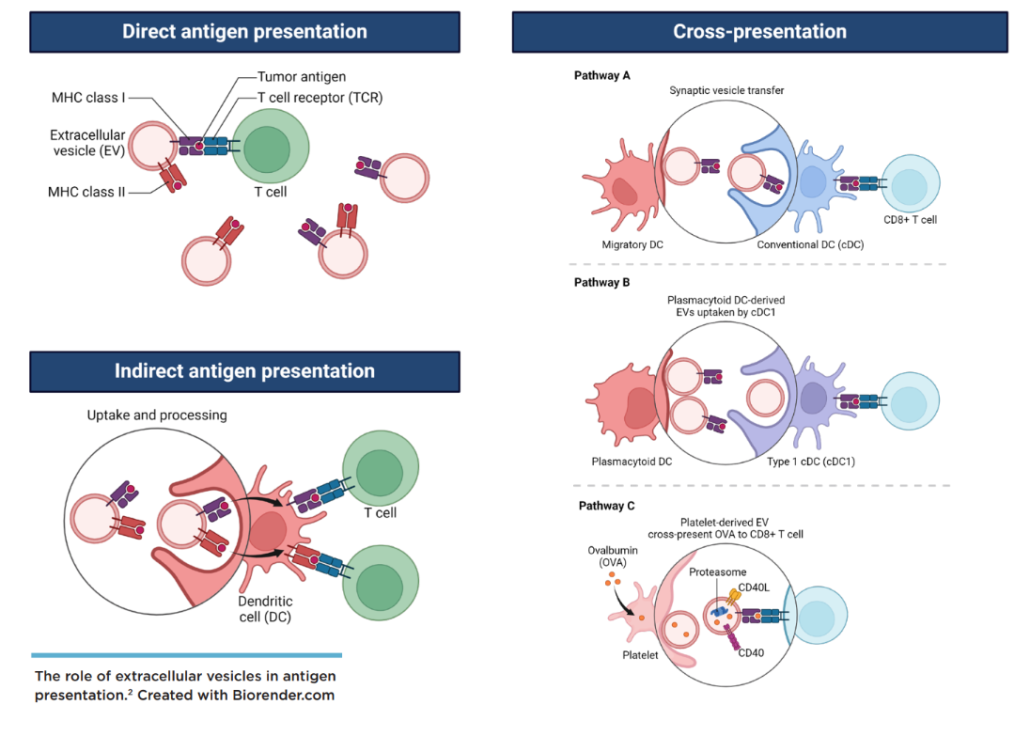
Figure 2. The role of extracellular vesicles in antigen presentation (Created with Biorender.com).
Risk of Disease Transmission
Using non-autologous exosomes is linked to cross- or intra-species contamination. Exosomes have been implicated in the transmission of infection, especially viral transmission, but also in other pathogenic infections, such as bacterial, fungal, and parasitic infections (Figure 3).5,6 Additionally, non-autologous exosomes carry the potential to introduce foreign proteins that may accelerate neurogenerative processes.7
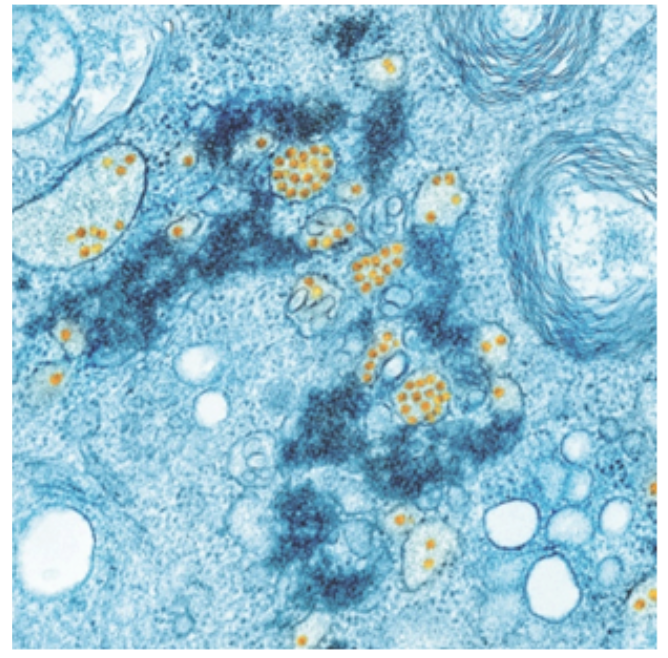
Figure 3. Infectious disease (Image of the National Institute of Allergy and Infectious Diseases [NIAID])
Exosomes Route of Administration
So far, no exosome-based products approved by regulatory agencies can be injected. On the other hand, all exosome-based products marketed as cosmetics must be topically applied. Only naïve autologous exosome-based products, such as those obtained with the MCT System, can be injected into the patient.
Types of Exosomes and Regulatory Aspects*
Based on their origin, exosomes can be classified into several categories:
- Naïve exosomes: Obtained from unmodified sources such as the autologous exosomes, which would be regulated similarly to cellular therapies.
- Naïve exosomes form genetically modified sources: These exosomes must be classified as biologics because the exosomes are unmanipulated.
- Modified exosomes from genetically manipulated sources: These exosomes must be considered under gene therapy regulations.
- Naïve exosomes from unmodified sources with synthetic content: These exosomes must be categorized as combination products with exosomes serving as delivery system.
*Must be verified according to the regulations of each country.
The long-term effects of the negligent use of cosmetics as injectable treatments and the consequences are still unknown.
References
1. Janouskova O, Herma R, Semeradtova A, Poustka D, Liegertova M, Malinska HA, et al. Conventional and Nonconventional Sources of Exosomes–Isolation Methods and Influence on Their Downstream Biomedical Application. Front Mol Biosci [Internet]. 2022 May 2;9. Available from: https://www.frontiersin.org/articles/10.3389/fmolb.2022.846650/full
2. Herrmann IK, Wood MJA, Fuhrmann G. Extracellular vesicles as a next-generation drug delivery platform. Nat Nanotechnol [Internet]. 2021 Jul 1;16(7):748–59. Available from: https://www.nature.com/articles/s41565-021-00931-2
3. Xia Y, Zhang J, Liu G, Wolfram J. Immunogenicity of Extracellular Vesicles. Advanced Materials [Internet]. 2024 Aug 27;36(33). Available from: https://onlinelibrary.wiley.com/doi/10.1002/adma.202403199
4. Buzas EI. The roles of extracellular vesicles in the immune system. Nat Rev Immunol [Internet]. 2023 Apr 4;23(4):236–50. Available from: https://www.nature.com/articles/s41577-022-00763-8
5. Chaudhari P, Ghate V, Nampoothiri M, Lewis S. Multifunctional role of exosomes in viral diseases: From transmission to diagnosis and therapy. Cell Signal [Internet]. 2022 Jun;94:110325. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0898656822000869
6. Xiong J, Ashraf U, Ye J, Cao S. Extracellular Vesicles in Pathogenic Infection, Transmission, and Immunity. Engineering [Internet]. 2024;43:228–40. Available from: https://www.sciencedirect.com/science/article/pii/S2095809924003801
7. Hartmann A, Muth C, Dabrowski O, Krasemann S, Glatzel M. Exosomes and the Prion Protein: More than One Truth. Front Neurosci [Internet]. 2017 Apr 19;11. Available from: http://journal.frontiersin.org/article/10.3389/fnins.2017.00194/full