What Is Autologous Platelet-Poor Plasma Gel and How Is It Obtained?
Autologous platelet-poor plasma (PPP) gel, also called plasma gel (PG) or albumin gel, is a protein-based blood-derived biomaterial prepared without utilizing the intrinsic coagulation pathway.1 PPP gel is produced when PPP is heated to 70–90°C, with the heating time adjusted to achieve the desired gel consistency. During this process, plasma proteins—mainly albumin—undergo irreversible denaturation, where they lose their secondary, tertiary, and quaternary structures and revert to their primary polypeptide chains. This process forms a cross-linked, biodegradable gel with tunable mechanical properties.2
Properties of Autologous PPP Gel
The PPP gel serves as a complementary formulation, providing volume and mechanical stability at the injection site.3 PPP gel is highly hydrophilic and capable of absorbing large amounts of water without dissolving, thanks to its cross-linked architecture; thus, it is also called “hydrogel.” These properties make them excellent carriers for extracellular vesicles, such as exosomes:
- Preventing premature clearance from the injection site.
- Maintaining a localized, concentrated dose.
- Providing sustained and more controlled therapeutic release.
Furthermore, the autologous nature of the gel means it is highly biocompatible and easily recognized by the host’s immune system as its own.
A New Strategy for Combining Autologous Plasma Gel and Exosomes
Meta Cell Technology has developed a novel approach for combining the traditional PG with the innovative preconditioned PRP using the MCT System that enhances the release of exosomes.4
- PPP Gel Formation: Using the Exo-Tech Dry Bath, we create a gel from platelet-poor plasma, thereby providing a stable and biocompatible scaffold.
- PRP Photothermal Biomodulation: Using the MCT Unit, we apply photothermal biomodulation to liquid-phase PRP to enhance the release of exosomes (the final product is referred to as MCT Exosomes from now on)*[1].
By combining these two phases, a PPP gel serves as the matrix, and photothermally biomodulated PRP acts as the bioactive component. This approach preserves the integrity of regenerative molecules while producing a gel with an intermediate consistency that combines the mechanical and biological benefits of both components (the final product, hereinafter referred to as Exo-Tech Gel).
Including exosomes in hydrogels, we obtain a two-phase exosome release:5
- An initial rapid release of surface-bound exosomes.
- A slower, steady “plateau phase” controlled by diffusion and hydrogel degradation.
How to Prepare the Autologous Combination
1. Sample Obtention:
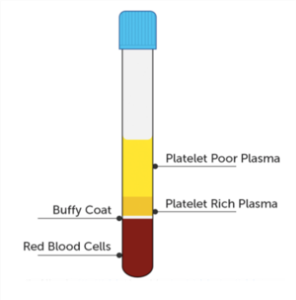 Withdraw the whole blood from the patient into a tub with anticoagulant and centrifuge it to separate components based on their density.
Withdraw the whole blood from the patient into a tub with anticoagulant and centrifuge it to separate components based on their density.
- Bottom: erythrocytes.
- Middle: “buffy coat” containing white leukocytes and platelets.
- Top: plasma (platelet-rich plasma and platelet-poor plasma).
2. Plasma Gel Formation:
Extract the PPP fraction within a syringe and place it inside the Exo-Tech device. Apply the selected heat up to 90ºC. Adjust the time to achieve a gel consistency adequate for the treatment procedure. The product obtained is the PG. Keep the sample in the syringe.
3. MCT Exosomes Obtention:
Extract the PRP fraction with a syringe and place it inside the MCT Kit. Load the MCT Unit and press the “Exosomes” preset. When the cycle is completed, remove the MCT Kit from the MCT Unit and extract the sample using a syringe.
4. Exo-Tech Gel Obtention:
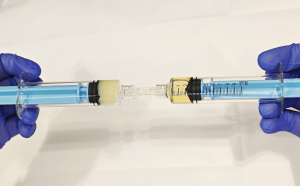
To obtain the final product, connect the two syringes: the PG (solid phase) and MCT Exosomes (liquid phase), and mix. The mixing produces a gel of intermediate consistency. This combination integrates mechanical stability with enhanced biological activity, resulting in optimized regenerative outcomes (Figure 2).
Key Advantages of This Approach:
Traditional exosome suspensions can disperse rapidly after injection, lowering their concentration.
Exo-Tech keeps exosomes at the injection site, improving concentration and prolonging effectiveness:
- Prolonged bioactivity5–8
- Enhanced localization5,8
- Prolonged tissue residence time5,8
- Synergistic regeneration7,9,10
- Autologous and safe11
References
-
Nakamura M, Masuki H, Kawabata H, et al. Plasma Gel Made of Platelet-Poor Plasma: In Vitro Verification as a Carrier of Polyphosphate. Biomedicines. 2023;11(11):2871. doi:10.3390/biomedicines11112871
-
Ong J, Zhao J, Justin AW, Markaki AE. Albumin‐based hydrogels for regenerative engineering and cell transplantation. Biotechnol Bioeng. 2019;116(12):3457-3468. doi:10.1002/bit.27167
-
Neinaa YME, Hodeib AAE, Morquos MM, Elgarhy LH. Platelet‐poor plasma gel vs platelet‐rich plasma for infraorbital rejuvenation: A clinical and dermoscopic comparative study. Dermatol Ther. 2020;33(6). doi:10.1111/dth.14255
-
Cordero L, Domingo JC, Sánchez-Vizcaíno Mengual E, Pinto H. Autologous platelet-rich plasma exosome quantification after two thermo-photobiomodulation protocols with different fluences. J Photochem Photobiol. 2025;29:100267. doi:https://doi.org/10.1016/j.jpap.2025.100267
-
Fan MH, Pi JK, Zou CY, et al. Hydrogel-exosome system in tissue engineering: A promising therapeutic strategy. Bioact Mater. 2024;38:1-30. doi:10.1016/j.bioactmat.2024.04.007
-
dos Santos RG, Santos GS, Alkass N, et al. The regenerative mechanisms of platelet-rich plasma: A review. Cytokine. 2021;144:155560. doi:10.1016/j.cyto.2021.155560
-
Jalowiec JM, D’Este M, Bara JJ, et al. An In Vitro Investigation of Platelet-Rich Plasma-Gel as a Cell and Growth Factor Delivery Vehicle for Tissue Engineering. Tissue Eng Part C Methods. 2016;22(1):49-58. doi:10.1089/ten.tec.2015.0223
-
Xie Y, Guan Q, Guo J, Chen Y, Yin Y, Han X. Hydrogels for Exosome Delivery in Biomedical Applications. Gels. 2022;8(6):328. doi:10.3390/gels8060328
-
Cavallo C, Filardo G, Mariani E, et al. Comparison of Platelet-Rich Plasma Formulations for Cartilage Healing. Journal of Bone and Joint Surgery. 2014;96(5):423-429. doi:10.2106/JBJS.M.00726
-
Cho EB, Park GS, Park SS, et al. Effect of platelet‐rich plasma on proliferation and migration in human dermal fibroblasts. J Cosmet Dermatol. 2019;18(4):1105-1112. doi:10.1111/jocd.12780
-
Doghaim NN, El‐Tatawy RA, Neinaa YME. Assessment of the efficacy and safety of platelet poor plasma gel as autologous dermal filler for facial rejuvenation. J Cosmet Dermatol. 2019;18(5):1271-1279. doi:10.1111/jocd.12876
[1] For more information about exosomes and MCT, read:
-
https://metacelltech.com/introducing-the-exosome-a-promising-new-player-in-regenerative-medicine/
-
https://metacelltech.com/autologous-vs-commercial-exosomes-how-to-face-current-market-challenges/